
| |
|
| | Research |
| |
| Oxidative stress plays an important role in the initiation and progression of many chronic diseases, including diabetes, cancer, and neurodegenerative diseases. Through the regulation of cytoprotective gene expression, the KEAP1-NRF2 stress response pathway is the principal inducible defense against oxidative and electrophilic stresses. |
|
| Under non-stressed conditions, the transcription factor NRF2 is targeted for ubiquitination and proteasome-dependent degradation by its negative regulator KEAP1, which forms an E3 ubiquitin ligase with CUL3 and RBX1. In response to a wide range of cellular stresses, the KEAP1-dependent E3 ubiquitin ligase is inactivated, which results in the stabilization of NRF2, and the upregulation of the antioxidant and cytoprotective gene expression programs. |
|
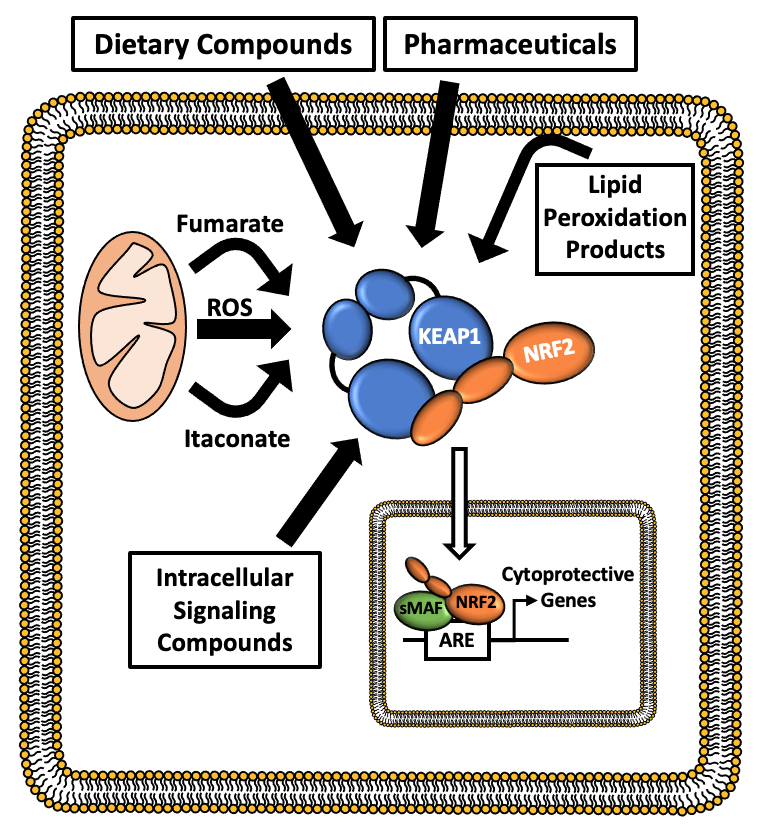 |
Mammalian models of medically important diseases utilizing Nrf2 knockout (KO) mice have directly implicated NRF2 activity in a wide range of human pathologies, including metabolic syndrome, diabetic nephropathy, rheumatoid arthritis, and Alzheimer’s disease. The inducible nature of the pathway means that, by clearly delineating the molecular mechanism of NRF2 activation, we can optimize the design and activity of NRF2 modulators in order to mitigate the deleterious effects of oxidative stress on human health. As such, a thorough understanding of the KEAP1-NRF2 pathway may lead to the development of novel treatments for a broad range of human diseases.
|
CGI-design
|

